THE REGENERATION REVOLUTION: Stanford’s Breakthrough in Cartilage Repair
STANFORD, CALIFORNIA — For millions, the prospect of an aging knee has long been a one-way street: pain, limited mobility, and the eventual, inevitable surgery for a joint replacement. However, new research published in early 2026 by a team at Stanford Medicine has introduced a potential paradigm shift. They have identified a method to reverse cartilage loss—not by adding external stem cells or performing surgery, but by “reprogramming” the cells that are already inside your knee.
The “Gerozyme” Inhibitor: Blocking the Aging Signal
The core of this discovery is a protein called 15-PGDH, which researchers have dubbed a “gerozyme” (an enzyme that increases with age).
As we age, levels of 15-PGDH rise in the knee, effectively acting as a chemical brake on the joint’s natural ability to repair itself. When this protein is active, it prevents the cartilage cells (chondrocytes) from maintaining their structural integrity, leading to the thinning and degradation we associate with osteoarthritis.
How It Works: Reawakening, Not Replacing
This is where the distinction regarding “no surgery or stem cells” becomes critical. Previous attempts at cartilage regeneration often involved transplanting external stem cells into the joint—a complex, expensive, and often inconsistent process.
This new approach is different:
Cell Reprogramming: Instead of bringing in new “construction workers” (stem cells), the researchers used an inhibitor drug to block the 15-PGDH protein.
The “Youthful” Shift: Once the protein was blocked, the existing chondrocytes in the joint “reawakened.” They shifted their gene expression back to a more youthful state, allowing them to naturally begin producing new, healthy cartilage.
The Result: In studies on aging mice and injured joints, the treatment didn’t just stop the degradation; it triggered the regrowth of thicker, functional cartilage.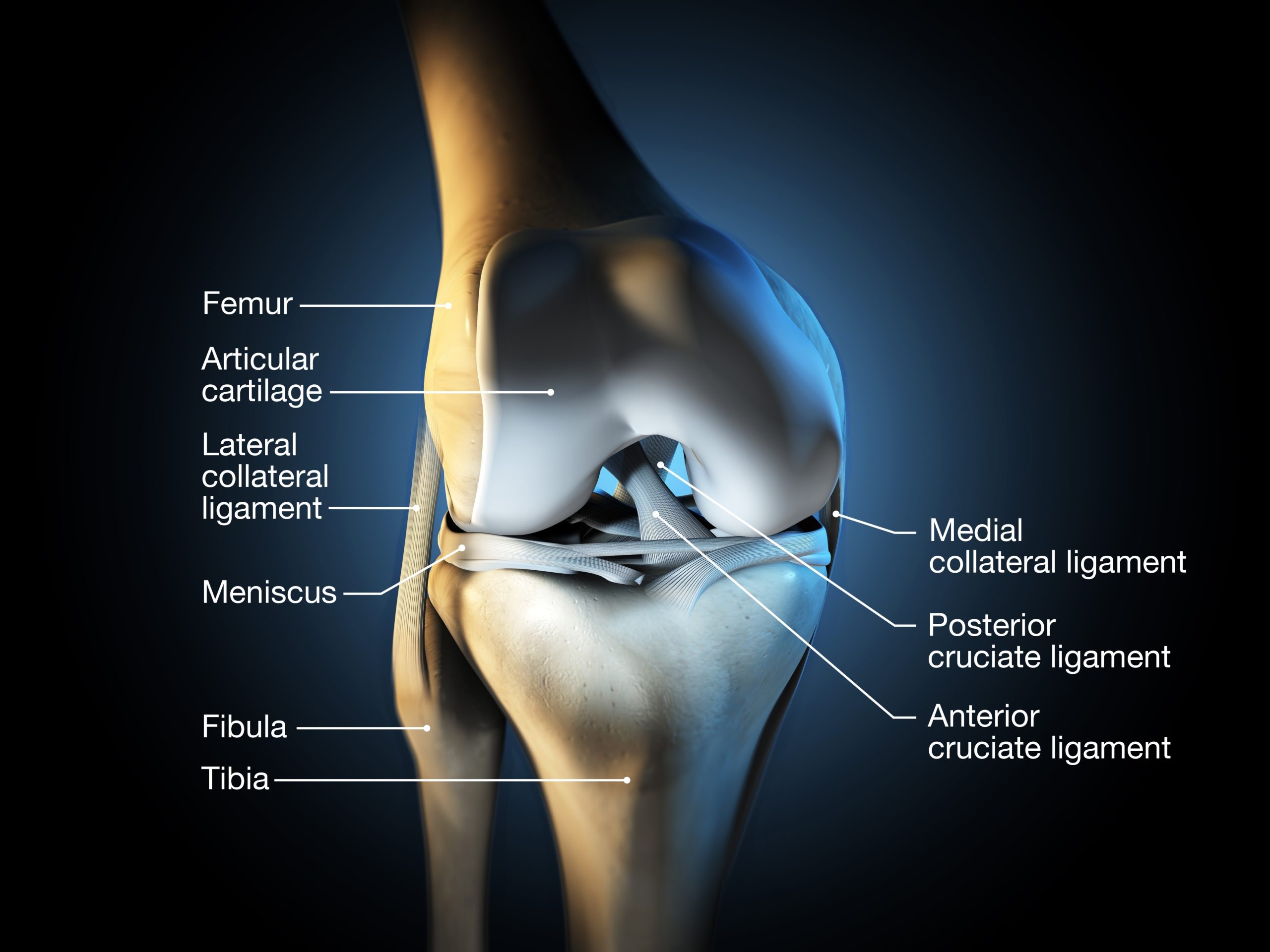
What This Means for Patients
While it is crucial to temper expectations—this has been successfully demonstrated in mice and in human tissue samples in the lab, but it has not yet completed full human clinical trials—the implications are massive:
The “Pill” Potential: Because the mechanism works by blocking a protein, it is theoretically possible that this could eventually be delivered via a local injection or even an oral medication, rather than a surgical procedure.
Proactive Protection: The study also showed that the treatment could prevent the development of arthritis after injuries (like ACL tears), potentially saving athletes and active adults from the long-term damage that usually follows those injuries.










